The regeneration of bone defects in the lower jaw is an important topic, but with few therapeutic alternatives.
Planning the future restoration of the lower jaw area, a correct diagnosis of the defect and adequate case planning are essential.
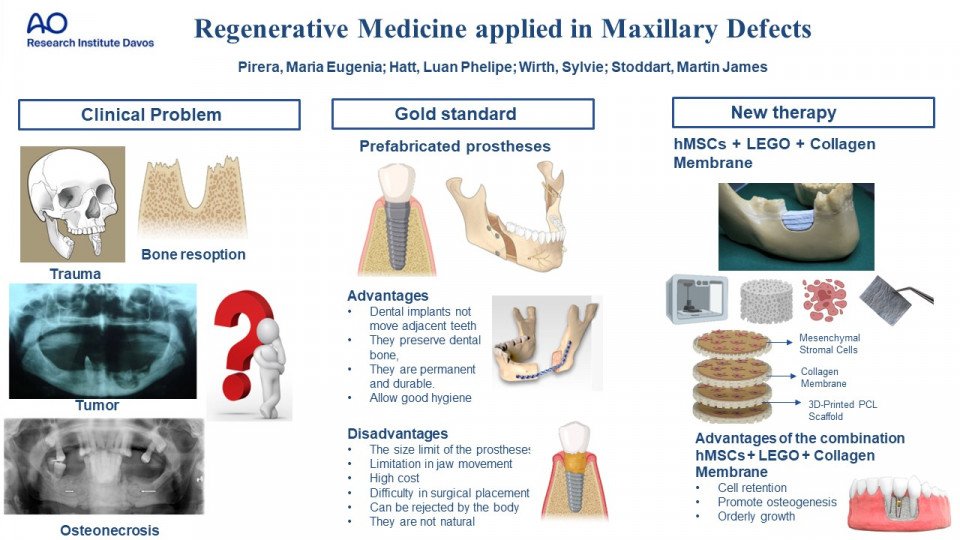
There are several causes of bone defects, although a simple tooth extraction and periodontopathies are the main diseases that cause this clinical problem. Additionally, on a smaller scale, other conditions such as infections, trauma, tumors, cysts and even the use of dental prostheses can increase bone resorption leading to great deformities.
When teeth are extracted, the bone that supports them tends to shrink over time. This process is called resorption and is a natural consequence of the lack of stimulation to the bone by the forces exerted on the teeth. Resorption of the alveolar bone (the bone that supports the teeth) begins almost as soon as the tooth is extracted and continues over time. The bone loses both height and width through resorption. When multiple teeth are lost, with or without dentures to replace them, there can be significant bone loss in the jaw, and this can lead to difficulty in subsequent rehabilitation.
Guided Bone Regeneration (GBR) is currently considered a therapy of great importance in Implantology, to promote bone regeneration in bone defects of the jaw. The purpose is to create a suitable area for subsequent rehabilitation with implants or conventional prostheses. GBR is based on the use of resorbable and non-resorbable membranes in combination with filler biomaterials such as bone from various sources or alloplastic materials with mechanical barrier functions, tending to exclude epithelial and conjunctival cells from the repair area, allowing the invasion of osteoprogenitor cells.
Scientific evidence shows that resorbable (biocompatible plastics) membranes can be successfully used for GBR.
However, in certain clinical situations, these membranes do not resist the pressure exerted by the tissues during healing, therefore, their use is advised in combination with different types of grafts, with the intention of maintaining these structural spaces.
3d printer scaffolds are fundamental components for tissue engineering. but since there are still challenges to overcome, such as the correct combination of their mechanical properties, degradation speed, porosity, biocompatibility and functional integration with the bone, these must be combined with other materials.
Due to the printing porosity and low attachment properties of the scaffold cells that are seeded on top fall through and cannot be retained as previous studies have showed.
To provide different alternative to existing treatments, the present work aims to find a suitable collagen membrane capable of retaining the cells within the scaffold structure previously designed with a configurable layer composition based on the LEGO® principle to achieve the promotion of cell viability and response.
The LEGO®-like structures are virtually designed using CAD software (Autodesk Fusion 360®) and printed into layered scaffolds composed of polycaprolactone (PCL) Using a RegenHu 3D Discovery® bioprinter. The collagen membranes that we used were: 1. Lyostypt (B. Braun) and 2. Collagen Cell Carrier (CCC) (Viscofan Bio Engineering). These membranes are placed in between layers of the 3D-printed scaffolds layers that interlock with each other via the LEGO® system. We seeded mesenchymal stromal cells (hBM-MSCs) on top of the assembled scaffold. In a 7-day retention experiment we examined the viability of hBM-MSCs within the scaffold with and without collagen membranes at two-time points Day 1 and Day 7. We performed DNA and LIVE/DEAD assays and we were able to observe a good cell survival showing 1. that membranes can retain cells, which was not possible testing the scaffold only 2. that hBM-MSCs on CCC membranes showed increased spreading compared to the Lyostypt membrane.
We can show that hBM-MSCs can be retained by the combination of the collagen membranes and the LEGO® inspired interlocking system and opens a wide possibility of achievements in reconstructive medicine. We propose it as a new therapeutic alternative in the tissue engineering approach for reconstruction of jawbone defects. Ongoing tests aim to demonstrate the osteogenic capabilities of collagen membranes placed inside 3D printed scaffolds.