Introduction:
Normal tissue repair, and integration of implanted medical devices, involves a controlled immune response. The recruitment of cells to remove damaged tissue occurs in the early phase, before switching to a later healing phase where cells such as M2 macrophages support tissue regeneration. In case of excessive or chronic inflammation at the injury site (e.g. due to infection or underlying systemic inflammation), this process is disturbed, slowing down tissue repair.
Immunomodulatory treatments aim at lowering inflammation for these difficult cases, however orally administered or intravenously injected anti-inflammatory drugs may have off-target effects due to systemic administration. Local application could overcome these drawbacks by targeting the injured site directly. In this project, we produced and characterized a bacterial exopolysaccharide, a type of sugar produced by bacteria, from a Bifibobacterium (BIEPS) as a potential coating for implanted devices.
Methods:
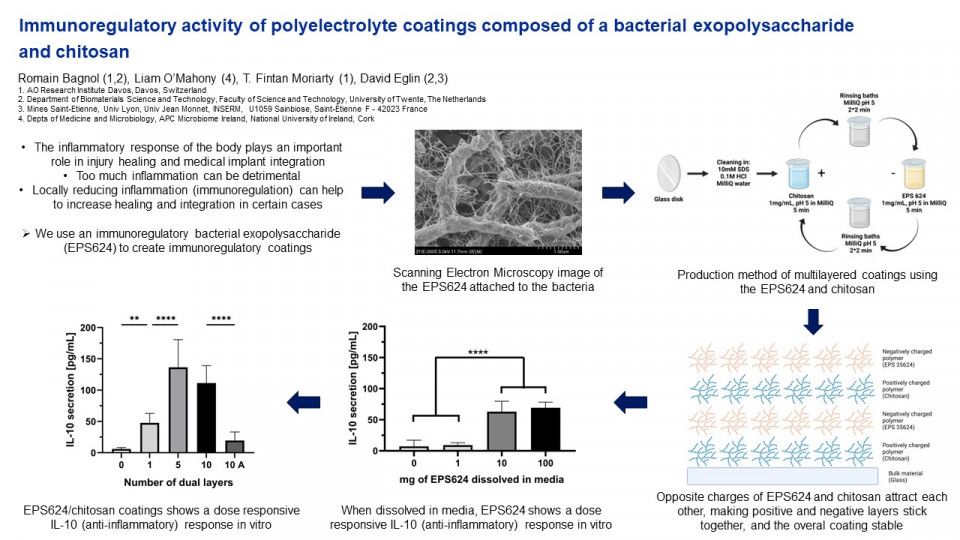
The BIEPS used for all experiments was isolated from. Bifidobacterium Longum subsp Longum. The production process consisted of bacterial growth on agar plates, collection of the bacterial biomass with a cell scrapper, resuspension and centrifugation of the bacterial biomass in PBS, filtration and precipitation of the supernatant with ethanol, dialysis and finally reverse phase column filtering of the precipitate. Polyelectrolyte multilayers were created from alternating layers of negatively charged BIEPS and positively charged chitosan on glass disks substrates. The process consisted of dipping the disks in baths containing 1mg/mL BIEPS at pH 5 or 1 mg/mL chitosan at pH5. Dipping was done for 5 minutes, with 2 rinsing baths in between each coating in a bath composed of MilliQ water at pH 5 for 2 minutes. A negative control replaced the BIEPS bath with an alginate bath, at concentration 1mg/mL and pH 3, with the rinsing baths also at pH3.
A zeta-sizer was used to measure zeta potential and hydrodynamic radius was measured by dynamic light scattering for the BIEPS, chitosan and alginate. Surface topography measurements through atomic force microscopy were used to characterize the coatings thicknesses and surface properties. Human Interleukin-10, an immunoregulatory cytokine secreted by cells such as T helper cells, macrophages, monocyte and dendritic cells, was quantified through ELISA to evaluate the immunoregulatory effect of coatings. Coated disks were exposed in vitro for 24h to human peripheral blood mononuclear cells.
Results:
The thickness of the coatings was found to be proportional to the number of layers deposited, with very thin coatings in the nanometer range. Zeta potential, quantifying polymer particle charges in solution, was measured for the BIEPS, chitosan and alginate at concentrations ranging from 0.01 mg/mL to 10 mg/mL. For all groups the absolute zeta potential value increased with polymer concentration. Dynamic light scattering was used to measure the hydrodynamic radius, a measure of the size of polymer particles in solution, for the BIEPS, chitosan and alginate at concentrations ranging from 0.01 mg/mL to 10 mg/mL.
When dissolving the BIEPS in media, an increase in IL-10 expression in hPBMCs was observed in a dose dependent manner. The control [6.27 ± 2.67 pg/mL] and 1µg/mL group [7.20 ± 2.17 pg/mL] had a similar expression, while a large increase was observed for 10µg/mL [79.68 ± 9.23 pg/mL] and 100µg/mL [105.66 ± 0.14 pg/mL]. Compared to the uncoated glass disk control [9.04 ± 1.12 pg/mL] IL-10 expression increased for all polyelectrolytes groups as the number of layer increased from 1 layer [41.35 ± 5.74 pg/mL], to 5 layers [90.93 ± 24.65 pg/mL], to 10 layers [116.57 ± 15.92 pg/mL]. For the 10 layers group, the IL-10 expression dropped to [18.17 ± 4.83 pg/mL] when the BIEPS was replaced by alginate, another negatively charged polymer, confirming the specific immunoregulatory action from the BIEPS.
Conclusion:
Our study could confirm the immunoregulatory property of BIEPS in vitro when incorporated in polyelectrolyte BIEPS/chitosan coatings. Future steps include further characterization of additional inflammatory cytokines to better understand the overall immune response to the BIEPS, ellipsometry measurements to determine the thicknesses of individual chitosan/BIEPS layers and size exclusion chromatography to determine the molecular weight and polydispersity of the polymers. These polyelectrolytes coatings could have potential for dampening inflammation and facilitating tissue healing.