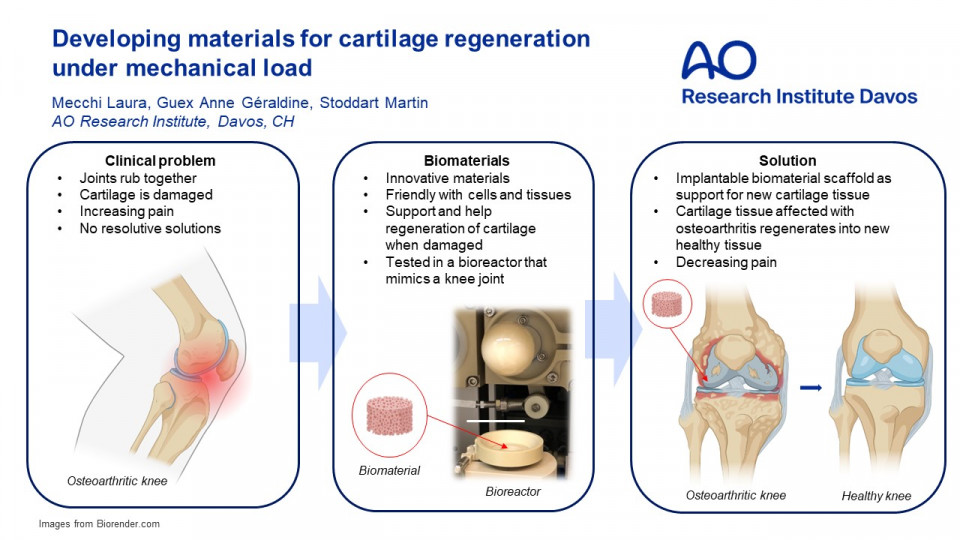
Introduction:
Osteoarthritis (OA) is one of the most common causes of disability in older adults, with increasing prevalence due to population ageing. It is a degeneration of the cartilage tissue in the joints, especially the knee, and it leads to increased pain during movement and a reduced range of motion. The main problem is that cartilage, unlike other tissues such as skin, does not regenerate easily. The current solutions for OA are anti-inflammatory drugs but, in the worst cases, the joint must be replaced with an artificial one by surgery. Thus, research on cartilage regeneration is increasing. Animal models are most accurate when developing cartilage therapies as they consider the three-dimensionality of the joints and their complexity closely resembles the complexity of the human body. On the other hand, the 3Rs principle encourages the reduction in the use of animals. Classical in-vitro models would then be an alternative: they are easier to reproduce and analyse. Their main drawback is that they are often too simplistic to describe the problem in a representative way. In between the previously mentioned models, the use of dynamically loaded in-vitro models offers the desired solution. In this model, a bioreactor that mimics the knee joint movement and a 3D scaffold, are used to study the cartilage regeneration mechanisms. The bioreactor is composed of three main units: a ceramic ball, a holder, and a motor. The scaffold is placed in the holder, against which the ceramic ball is pressed. The motor rotates the ball and moves it up and down to apply load, mimicking the motion of the joint. The scaffold can be made of various biomaterials that should mimic the properties of the joint cartilage. The gold standard for this model is using a polyurethane (PU) foam scaffold: PU is a polymer that presents the proper characteristics to mimic cartilage. The main drawback is its low reproducibility. The aim of this research is to design a scaffold whose properties mimic cartilage without sacrificing reproducibility. To address this issue, two approaches are being attempted: soft gels called interpenetrating polymer networks (IPNs) and 3D printed scaffolds, exploring new biomaterials use and new architectures.
Methods:
IPNs are prepared as reported elsewhere [1]. Different formulations are tested, to check for their gelation time. Samples are tested for swelling up to 7 days, for unconfined compression and for bioreactor loading in the absence of cells. The bioreactor loads in compression and shear for 6 hours. For the swelling test, the samples are covered evenly with PBS, incubated at 37°C and checked at different time points (24 hours, 5 days, 7 days).
Results:
Not all the formulations of the IPNs gelled in the expected time. Once gelled, they were too brittle to load in the bioreactor. These differences influenced the mechanical properties of the samples. All the samples showed brittle-like behaviour. The samples did not show a significant degree of swelling over the 7 days test and they retained their brittle-like behaviour over that period of time. Tests on the mechanical properties and the bioreactor loading are still ongoing, as well as production and tests on 3D printed scaffolds.
Conclusions:
IPNs need to be further tested to gain more data about their properties. Current and future experiments using the 3D printed technology could be the solution to obtain a reproducible scaffold which shows cartilage-like properties. Materials to be tested are PU [2] and polycaprolactone (PCL) [3]. Different architectures are also to be tested, in order to tune the mechanical properties of the scaffold to match the ones of interest.
References:
1. Jeon, O., et al., Highly Elastic and Tough Interpenetrating Polymer Network-Structured Hybrid Hydrogels for Cyclic Mechanical Loading-Enhanced Tissue Engineering. Chemistry of Materials, 2017. 29(19): p. 8425-8432.
2. Li, Z., et al., Mechanical load modulates chondrogenesis of human mesenchymal stem cells through the TGF-beta pathway. J Cell Mol Med, 2010. 14(6A): p. 1338-46.
3. Zimmerling, A., et al., 3D printing PCL/nHA bone scaffolds: exploring the influence of material synthesis techniques. Biomater Res, 2021. 25(1): p. 3.