Nowadays, diagnostics tests are usually made by the analysis of one or more biomarkers at the hospital. It typically takes more than one day to obtain the results. Moreover, specialized personnel and bulky, expensive instrumentation is required to perform the test. An alternative to this procedure involves the use of point-of-care (POC) analysis devices, that allow to achieve an accurate and specific analyte detection in the most convenient and rapid way for the patient. The present work describes the development of a novel POC platform for the quantification of biomarkers, based on an electrochemical immunoassay. This platform is based on screen-printed electrodes (SPEs) which can be miniaturized for integration in a compact and cost-effective POC device. This technology can achieve sensitivity and specificity that match the specifications of the standard methods.
One of the main challenges with the immobilization of antibodies on a sensor surface involve the number of steps necessary, which directly impacts the time required for the functionalization and the reproducibility of the processes in view of mass production. In order to bring a solution to such challenges, we propose a photoreactive polymer, which is capable to crosslink various categories of biomolecules onto the sensor surface, e.g. antibodies, enzymes, and receptors, by one-step UV activation.
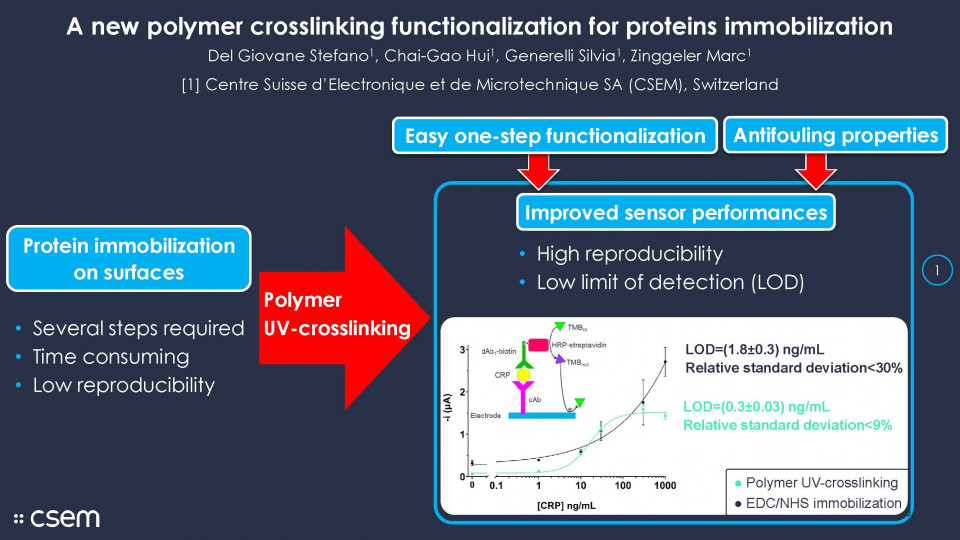
As a model system, we developed a C-reactive protein (CRP) sensor based on a sandwich immunoassay with a LOD<1 ng/mL. First, the polymer (Fig.1a) was drop-casted on the working electrode and immobilized by partial UV crosslink, then the capture antibody (cAb) was applied on the surface and covalently bound to the polymer backbone by UV crosslinking. Successively the entire sensor was incubated with bovine serum albumin, to block any sites that may be still available for unspecific binding.
For comparison, another sensor was prepared by immobilization of the antibody by EDC/NHS functionalization. The sensor (EDC/NHS) was prepared as illustrated in Fig.1b. The number of steps involved in this process is significantly higher than for the polymer-based sensor (Fig 1a), which we believe being the source of the decreased reproducibility of the sensor.
A biotinylated mouse anti-CRP IgG was used as the detection antibody (dAb1-biotin), then horseradish peroxidase-labelled streptavidin (HRP-strept) was bound as electrochemical tracer. The electrochemical reaction occurs by adding the 3,3',5,5'-tetramethylbenzidine (TMB) substrate after the immunoassay and the current is measured by chronoamperometry, by the reduction of the product (Fig. 3).
To characterize the antibiofouling effect, the biotinylated goat anti-mouse IgG (dAb2-biotin) was directly bound to the immobilized capture antibody (Fig. 2a). To measure the signal-to-noise ratio and characterize the anti-fouling properties, a fixed concentration of cAb was crosslinked on the electrode and the blank was prepared by crosslinking the same concentration of BSA. The results (Fig.2b) showed the antifouling effect of the polymer, the blank signal is equal to 0.12±0.06 µA even at higher concentrations of HRP-strep, since the non-specific binding is supressed by the polymer.
The immunoassay demonstrated that the immunoactivity of the cAb immobilized on the polymer is preserved and by consequent so is the capability of the system to detect CRP in buffer. The calibration curve gives a LOD of 0.30±0.03 ng/mL, while the IC50 is 16±4 ng/mL (Fig. 3). In comparison, the EDC/NHS sensor gained a higher dynamic range but presented a higher LOD 1.8±0.3 ng/mL. The comparison of the reproducibility shows that the relative standard deviation (RSD) of the polymer-based sensor is comprised between 5 and 9%, which is lower than the EDC/NHS sensor, ranging from 6 to 30%. The higher RSD is probably due to the higher number of steps for the preparation of the sensor.
In summary, the proposed polymer showed the capability of crosslinking biological sensing material to the sensor surface in one step by UV radiation, and an efficient antifouling behaviour. The comparison with EDC/NHS immobilization technique proved, a reduced time of functionalization a reduced number of processing steps, an increase of reproducibility and a decrease of the LOD. The next development steps will be to optimize the sensing for detection in human serum and the integration of the sensors into a multi-biomarker assay platform.
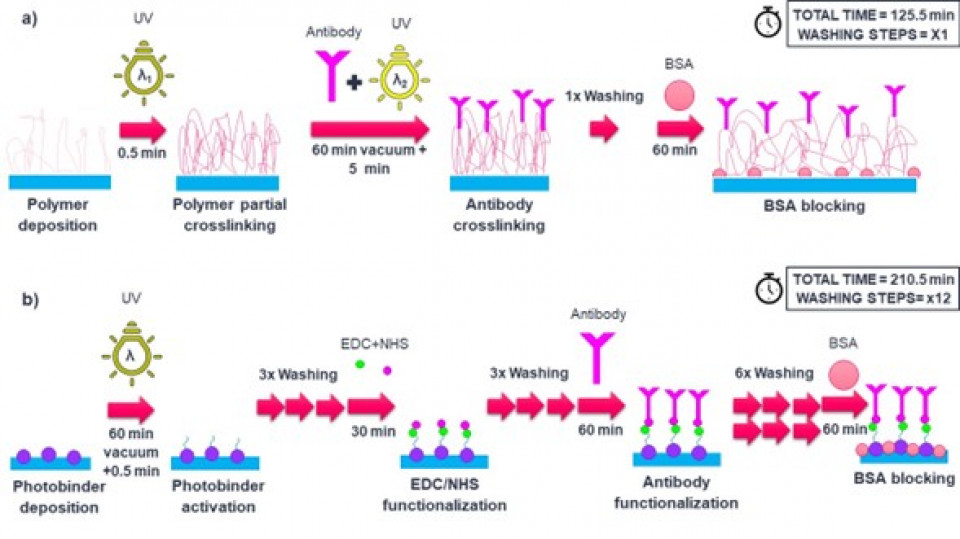
Figure 1. Sensor preparation: (a) the sensor surface functionalization with the photoactive polymer, and (b) with EDC/NHS chemistry.
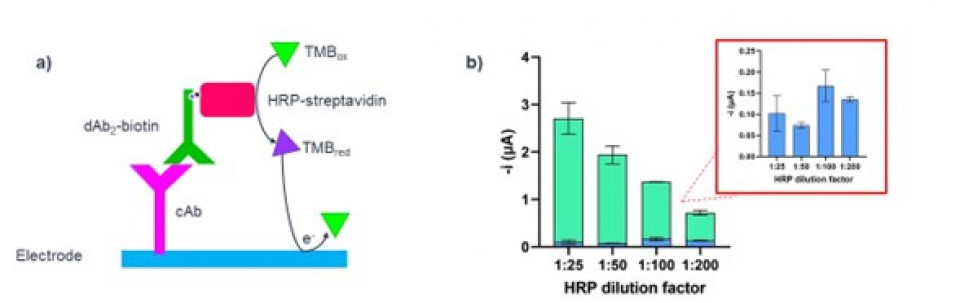
Figure 2. (a) Schematics of the immunoreaction and chronoamperometric detection used in the antifouling tests. (b) The antifouling property of the polymer is tested by incubation with different dilutions of HRP-Streptavidin. The blank signal (blue) is almost constant with increasing concentrations of HRP-Streptavidin, while the analyte signal (teal) increases. The experiment was conducted in buffer. N=3
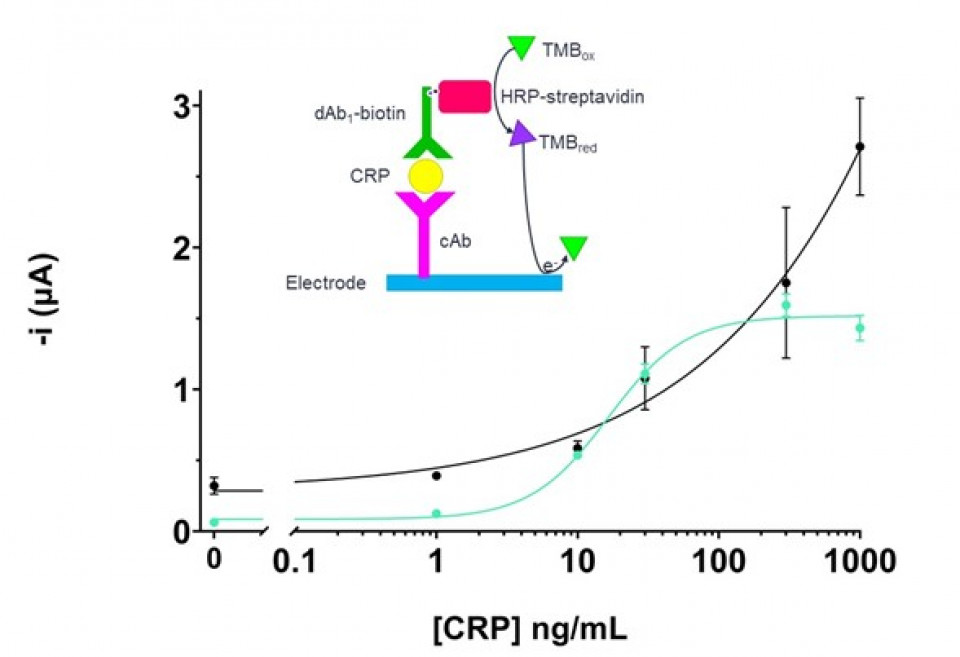
Figure 3. Schematic view of the CRP immunoassay and respective calibration curves, obtained by the polymer sensor (teal) and with the EDC/NHS sensor (black). The polymer sensor shows a lower LOD and an enhanced reproducibility. The experiment was conducted in buffer. N=3